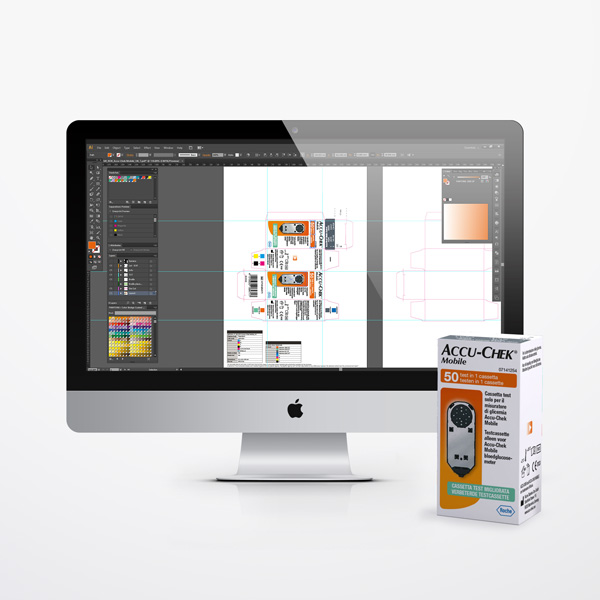
We create
artwork for
pharmaceutical
boxes, leaflets
and blisters.

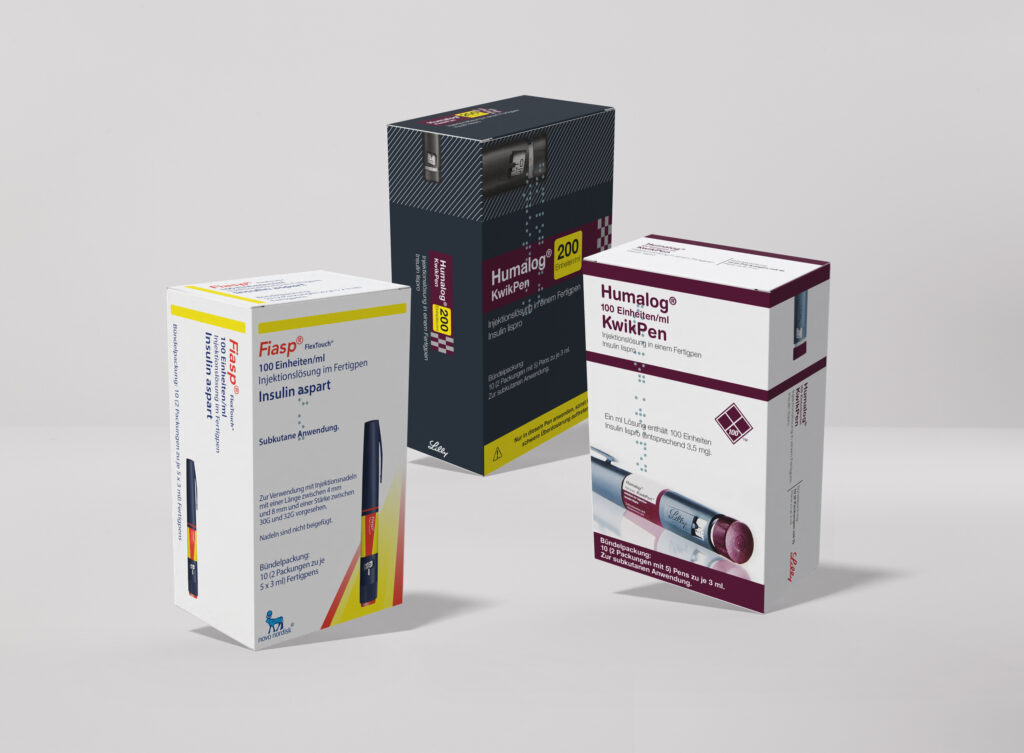

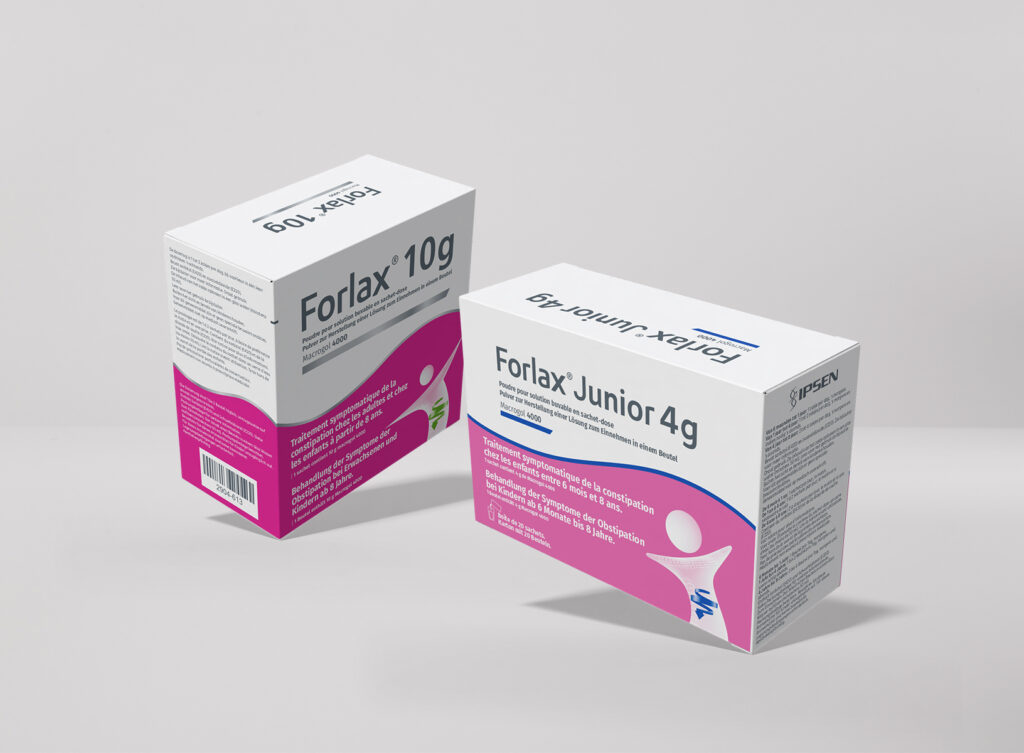

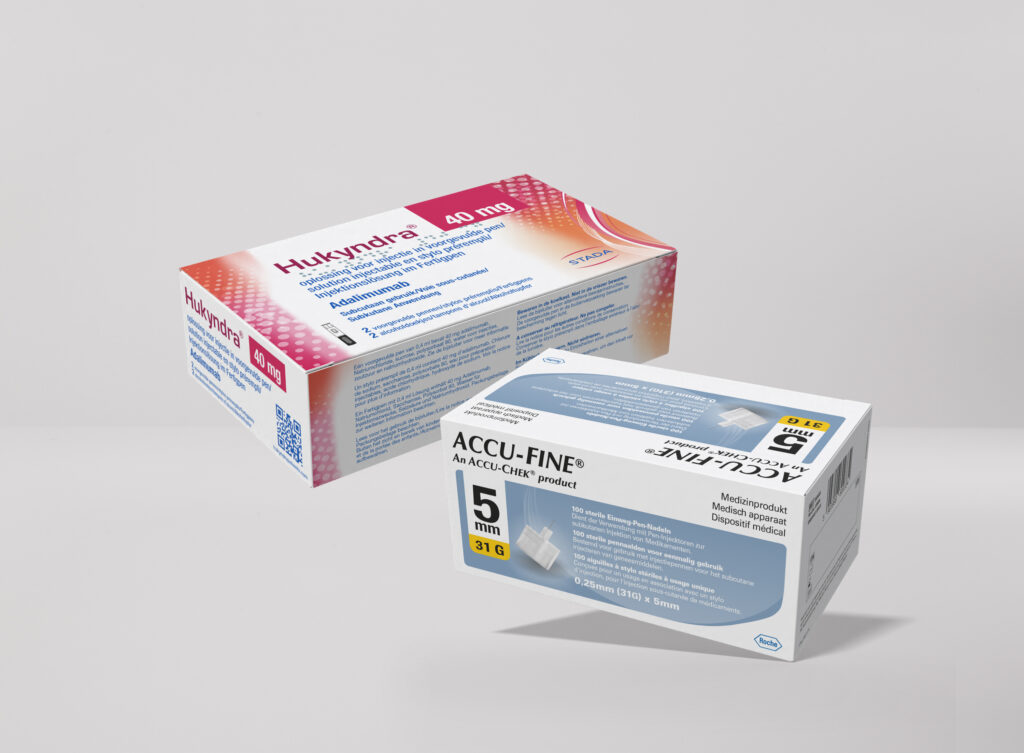
< Click and drag to see more >
WHO we are
With over 20 years of professional experience creating high-compliance pharmaceutical artwork for the European market, we deliver industry-leading results every day. From our strategic hubs in Leuven, Belgium, and Belgrade, Serbia, we serve as the premier specialist in creating pharmaceutical packaging, leaflets, and blisters for your brand.
We offer unique expertise in redrawing pharmaceutical packaging for parallel import, ensuring that your products meet all local language and regulatory requirements across different European jurisdictions. We manage the entire process, moving seamlessly from initial design to ready-to-print (RTF) files.
WHAT we do
Packaging design
for parallel-import boxes
From a plain scan, we create vectorized packages that are fully customizable and print-ready for your local market.
EMA
Application
Preparing fully digital submission documents for EMA, including 3D simulation views and necessary details.
Technical die cuts
Customised die-cut designs, tailored to your packaging requirements.
Package design
Designing or redesigning packaging that meets your needs and requirements.
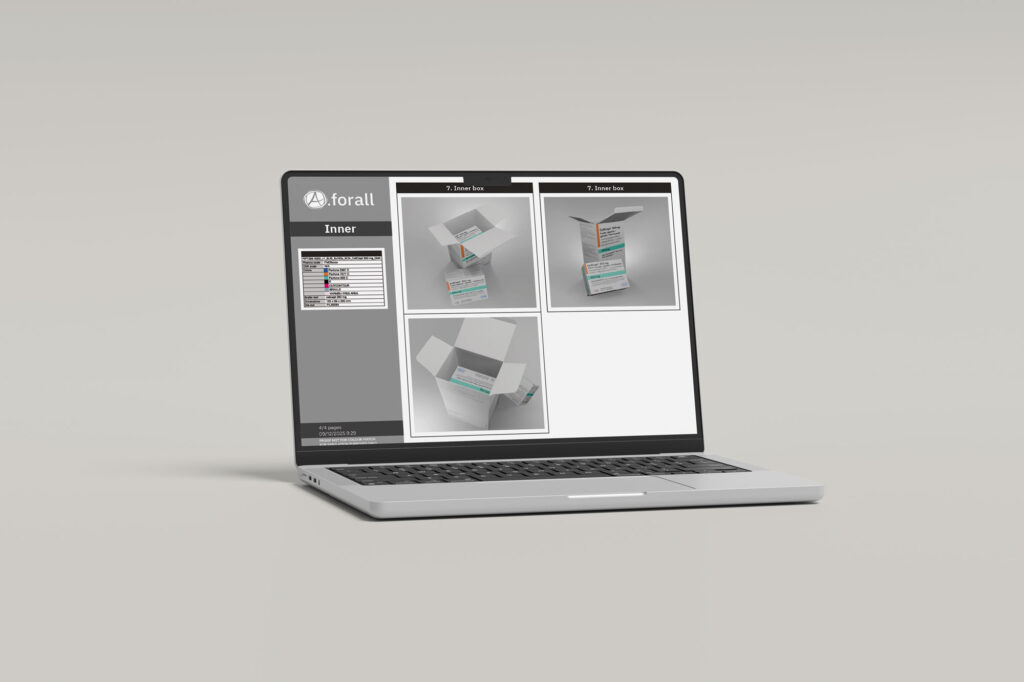
3D mockups
Realistic visualizations of your packaging before production or EU distribution approval.
Prototype boxes
Pre-production models for internal testing, inspection and regulatory submission.
Prepress preparation
Ensuring files are optimised and ready for offset or digital printing.
Fast communication
We respond quickly and efficiently to all requests.
EU distribution complience
We help you stay compliant with all legal requirements for distributing pharmaceutical products within the European Union.
Parallel Import Compliance
Navigating the complexities of parallel importation requires specialist knowledge and meticulous attention to regulatory detail. Whether a product is being repackaged, relabelled, or adapted for a new market within the European Economic Area, our team ensures that every requirement is fully met. This includes language compliance, mandatory safety information, traceability, and packaging integrity. We work in accordance with the standards set by authorities such as the FAGG, ANSM, and BfArM, ensuring your product reaches its target market legally, efficiently, and without compromise to patient safety.
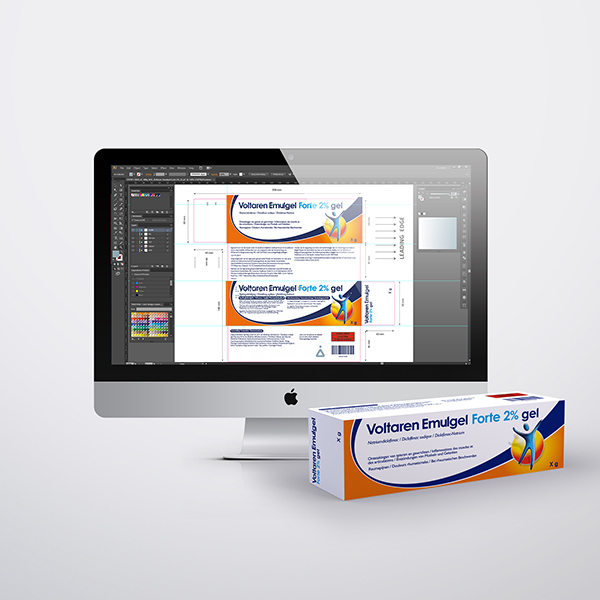
Packaging Artwork
Pharmaceutical packaging artwork is one of the most highly regulated and detail-critical disciplines in the industry, and we treat it as such. From cartons and labels to blister foils and overwraps, we develop artwork that is fully compliant with the requirements of the EMA, FAGG, CBG, and BfArM, while maintaining a clean and professional aesthetic that reflects your brand. Our structured review and approval workflows ensure that every text element, regulatory detail, and visual component is accurate, complete, and ready for market.
Creating Leaflets
The patient information leaflet (PIL) is far more than a regulatory obligation. It is a direct communication between your product and the patient. We develop leaflets that comply fully with the approved Summary of Product Characteristics (SmPC) and meet the readability and formatting standards required by authorities including the FAGG, ANSM, and EMA, including user readability testing where mandated. Whether your product is destined for a single market or requires multilingual adaptation across Belgium, the Netherlands, Germany, France, … Our team ensures every leaflet is precise, compliant, and clearly structured from the first draft to final approval.
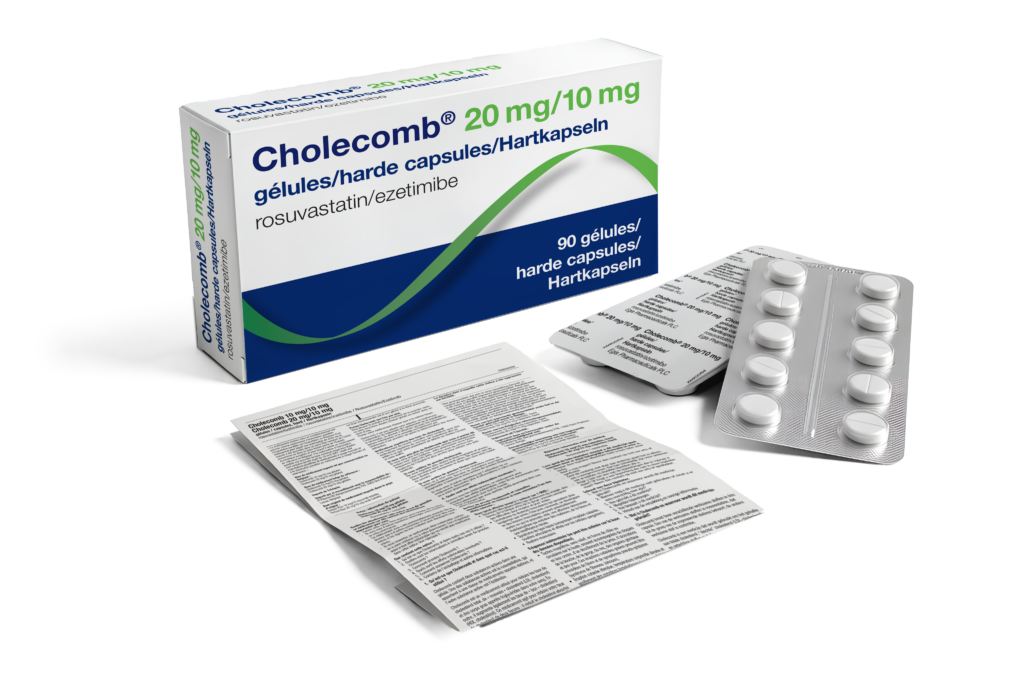
Blister Setup
A correctly configured blister is fundamental to product protection, regulatory compliance, and patient convenience. We manage every aspect of blister setup, including cavity dimensions, forming films, lidding materials, perforation patterns, sealing parameters, and the integration of braille, batch coding, and serialisation data. Our team works in full alignment with the technical and regulatory expectations of authorities such as the FAGG, BfArM, and IGJ, ensuring the final configuration meets all applicable market requirements. The result is a blister solution that is reliable, compliant, and optimised for shelf life.
3D Simulations
Before a single physical prototype is produced, 3D simulation allows your packaging to come to life in photorealistic detail. Our three-dimensional renderings give regulatory, marketing, and design teams a true-to-life view of cartons, labels, blisters, and other packaging components. This enables thorough evaluation of layout, colour accuracy, and text placement at an early stage, significantly reducing the risk of costly revisions later in the process. It also supports faster internal approvals, stakeholder presentations, and submissions to authorities such as the EMA and national bodies including the FAGG and ANSM, making it a smarter and more efficient way to bring compliant packaging to market.
EMA Application
The EMA (European Medicines Agency) region encompasses a broad and complex landscape of regulatory frameworks, language requirements, and market-specific submission standards. From centralised procedures managed by the EMA to national authority submissions handled by the FAGG, CBG, BfArM, and ANSM, our team has the expertise to manage your application with precision and strategic oversight. We prepare and coordinate all required documentation, including country-specific labelling, patient information leaflets, and supporting dossiers, ensuring your product achieves timely market access across every target country, fully compliant and without unnecessary delay.
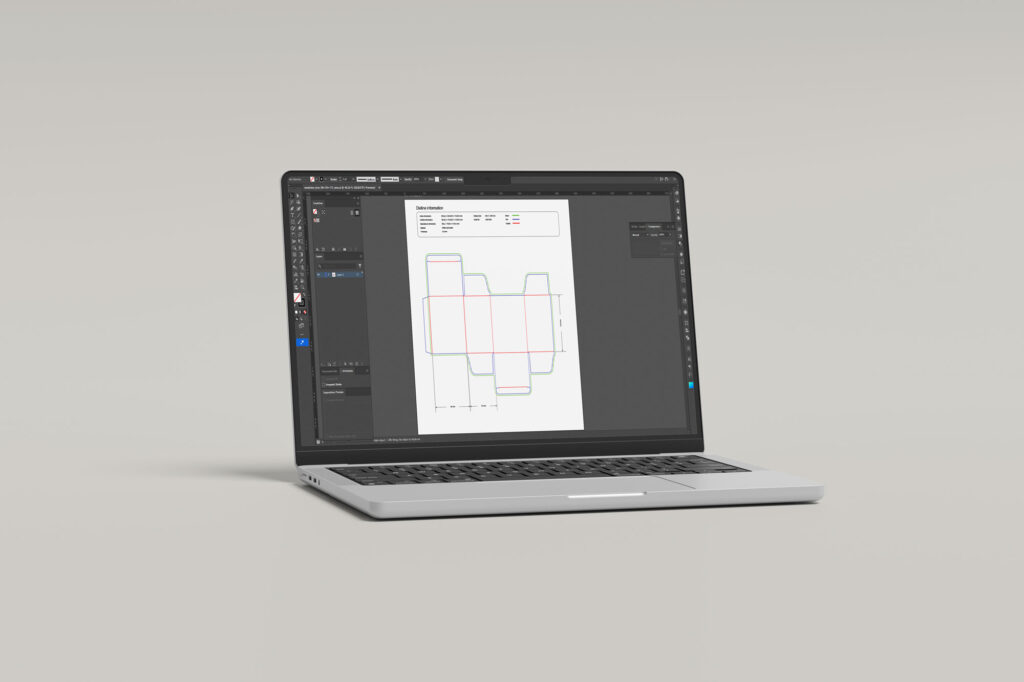
Diecut Drawings
Every folding carton, wallet, or leaflet begins with a precise and technically validated diecut drawing. This foundational document defines the exact dimensions, fold lines, cut patterns, and perforation specifications that determine how your packaging performs on the production line. We develop diecut drawings in close collaboration with printers, packaging engineers, and production teams, ensuring full compatibility with automated filling and assembly equipment and consistent manufacturing tolerances. Where applicable, diecut specifications are aligned with the packaging standards expected by authorities such as the FAGG and BfArM. Formal approval of the diecut drawing before artwork application is a critical quality step, and one we manage with the rigour that pharmaceutical packaging demands.
Prepress Artwork
Once your packaging design has been approved, our pre-press specialists will provide print files of the artwork. This includes colour separation, trap settings, image resolution verification, barcode validation, and font conversion, all conducted within a validated quality management framework. In the pharmaceutical industry, prepress is a zero-tolerance discipline. Any undetected error in a print-ready file can result in non-compliant packaging. Our team carefully examines every file to ensure that the approved version is reproduced accurately in print.
Prototypes With Braille
Physical prototypes incorporating braille (created with extra-thick UV spot varnish of 80 µm) provide the essential final verification that your packaging meets both regulatory requirements and the real-world needs of visually impaired patients. Under EU Directive 2004/27/EC, braille must accurately render the product name and strength on all pharmaceutical cartons, a requirement enforced across our core markets by authorities including the FAGG, IGJ, and ANSM. Prototype testing allows our team to confirm dot height, spacing, and tactile legibility against the applicable standards before production commences. By identifying and resolving any braille discrepancies at the prototype stage, we ensure your packaging goes to market fully compliant, thoroughly verified, and fit for every patient who depends on it.
WHY us
Complete Solutions
From concept to prototype, we provide end-to-end pharmaceutical packaging services across Europe. We manage everything from prepress and die-cuts to 3D mockups. We ensure your product is compliant with the regulations, modern, and fully market-ready for any European jurisdiction
Speed & Reliability
We combine technical precision with fast, responsive communication. We help you bring your pharmaceutical packaging to life quickly, meeting tight EU market launch deadlines without ever compromising on the rigorous quality standards your brand requires.
Trust & Expertise (20+ Years)
With over 20 years of proven experience in the European market, we specialize in pharmaceutical packaging and parallel import artwork. Our deep knowledge of local and international regulations ensures that your brand meets strict industry requirements while delivering standout design and superior quality that performs on the shelf.
"Turning strict standards into standout design."
Frequently Asked Questions
What do I need to start my new package?
For parallel-imported packaging:
- the dimensions of the imported packaging
- information about the local packaging
- details of the required changes
If required, we can provide a starter template based on established procedures, covering everything from initial set-up and corrections to final print files, to ensure that the briefing and process run smoothly and efficiently.
For new packaging and projects:
You only need a basic idea of what you would like to achieve. Any additional details you provide will enhance the final result.
What is your turnaround time for corrections?
Corrections are normally implemented within 24 hours. High-priority corrections are often implemented on the same day.
Can you create barcodes and die-cuts?
Yes, we can generate all types of barcodes and produce any die-cut designs you require.
Can you help with existing artwork?
Absolutely. We can adapt your current artwork. Based on the condition of your file, we’ll determine the best approach to refine or update it.
Can you make a 3D visualization of my product?
Yes, we can produce detailed 3D artwork to visualize your packaging before production.
Can you create a prototype for my box?
Yes, we can build a prototype based on our created design, including braille printed embossed.
Can I animate the created packaging?
Yes, we can fully animate our create package, including 3D mockups suitable for websites or online platforms.
Will my file be print-ready?
Yes, we ensure that your file is fully prepared and ready for print in offset or digital, depending on what you need.